Study of Anti-Cancer Property of Olive Oil Against DMBA Induced Breast Cancer in Rats
Farhat Yasmin1, Sidhnath Prasad Yadav Deen2, Shabana Kareem3, Tushar Kanti Maiti4, Pintoo Kumar Niraj4 , Mohammad Ali4*
1P.G., Department of Biotechnology, Magadh University, Bodh Gaya, Bihar, India
2P.G., Department of Zoology, Magadh University, Bodh Gaya, Bihar, India
3Department of Botany, A. N. College, J495+CX4, Boring Rd, Sri Krishna Puri, Patna, Bihar, India
4Research Department, Mahavir Cancer Sansthan, Phulwarisharif, Patna, J495+CX4, Boring Rd, Sri Krishna Puri, Patna, Bihar, India
*Corresponding Author: Mohammad Ali, Research Department, Mahavir Cancer Sansthan, Phulwarisharif, Patna, J495+CX4, Boring Rd, Sri Krishna Puri, Patna, Bihar, India.
Received: 19 March 2026; Accepted: 25 March 2026; Published: 7 April 2026
Article Information
Citation: Farhat Yasmin, Sidhnath Prasad Yadav Deen, Shabana Kareem, Tushar Kanti Maiti, Pintoo Kumar Niraj, Mohammad Ali. Study of Anti-Cancer Property of Olive Oil Against DMBA Induced Breast Cancer in Rats. Archives of Clinical and Biomedical Research. 10 (2026): 147-153.
View / Download Pdf Share at FacebookAbstract
Background: The present study has been designed to evaluate the tumor regression properties of olive oil in a rat model with DMBA-induced breast tumors.
Methods: Female Charles Foster rats were given DMBA orally (20 mg/mL diluted in olive oil). The rats received oral treatment with olive oil (2000mg/ kg) for four weeks after developing breast tumors.
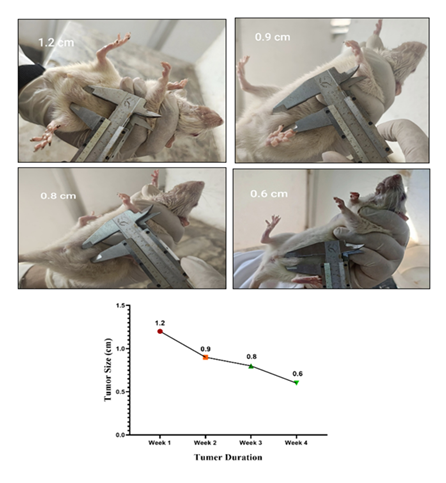
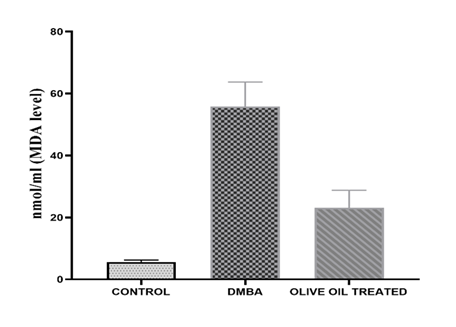
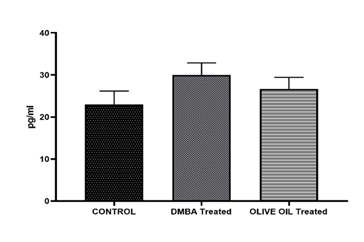
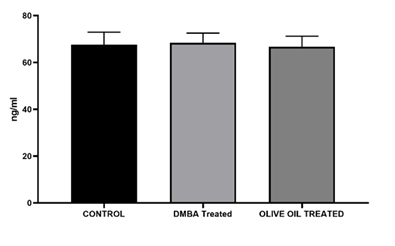
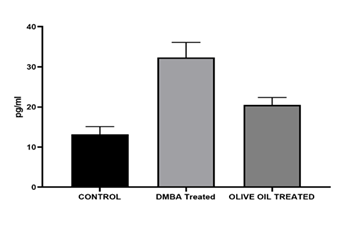
Results: In the present investigation after administration of olive oil for 4 weeks reduction in tumor size was observed from 1.2 cm to 0.6 cm associated with a significant decrease in LPO level. The decreased serum tnf alpha level was also observed as compared to DMBA treated. On the other hand, therapeutic response of olive was also observed in histopathological study. Apoptosis, reduced mitoses and inflammatory infiltration may reflect the anticancer potential of olive oil. A decrease in level of estradiol was observed as compared to DMBA treated, however no changes in serum progesterone level was observed.
Conclusion: Based on the complete study, it can be concluded that olive oil has anti-proliferative properties by slowing the growth of breast cancers in rat models.
Keywords
Rats; Anti-Cancer; Olive Oil; DMBA; Breast; Cancer
Article Details
1. Introduction
The management of breast cancer requires an approach that involves multiple disciplines. In the treatment of locally advanced breast cancer, triple-negative breast cancer, and tumours expressing HER2 (human epidermal growth factor receptor 2), mastectomy, followed by adjuvant chemotherapy, has been a treatment method that has been utilised rather frequently. Individuals who have been diagnosed with triple-negative breast cancer typically have unfavourable outcomes, and the treatment measures that are available to them are primarily restricted to systemic chemotherapy. Immunotherapy and antibody-drug conjugates are two promising methods that have the potential to change the existing landscape of breast cancer treatment. One of the most important aspects to take into consideration when developing a treatment strategy is the widespread excitement that surrounds newly launched pharmaceuticals, despite the fact that these medications frequently demonstrate insignificant or even nonexistent increases in survival outcomes. Therefore, making a careful selection is of the utmost importance, and the fact that it is cost-effective makes it particularly suitable for situations with little resources, such as countries like India.
Diet, body mass index, and physical activity are three lifestyle factors that have been shown to have a major impact on the incidence of cancer. These lifestyle factors have garnered a great amount of attention from the scientific community as modifiable cancer risk factors. As a result, a number of different dietary patterns have been linked to an increased risk of breast cancer, with dietary lipids being a prominent role in this correlation [1]. Despite the fact that clinical research into these correlations continue to be contested, laboratory studies have produced extensive evidence demonstrating the impact that dietary lipids have on breast cancer [2,3]. The total amount of dietary fat consumed as well as the particular type of fat that is consumed are both factors that influence the potential for modulation. Therefore, diets that are abundant in n-6 polyunsaturated fatty acids (PUFAs), particularly linoleic acid, have been shown to have stimulatory effects on breast cancer in animal models.
Mammary tumours in rodents can be caused by the chemical carcinogen known as DMBA, which is commonly used in practice. The disruption of tissue redox balance that occurs throughout the process of DMBA-induced carcinogenesis is what ultimately results in oxidative stress, which in turn causes biochemical and pathophysiological abnormalities in rats [4,5]. Through the process of lipid peroxidation, the production of reactive oxygen species is responsible for the initiation of cellular injury, which in turn causes changes at both the cellular and subcellular levels. It has been noticed that the incidence of cancers that are oestrogen receptor-positive has been on the rise, whereas the incidence of cancers that are oestrogen receptor-negative has been on the decline [6]. These cancers are impacted by oestrogen, which encourages the proliferation of cancer cells [7]. Oestrogen receptor-positive cancers are affected by oestrogen. The compound known as 7,12-dimethylbenz(a)anthracene (DMBA) is considered to be one of the most potent carcinogens that can cause cancer in the mammary glands of animals, including rodents (Median, 1982).
Within a mouse model that has been induced with DMBA to develop mammary tumours, the purpose of this study is to investigate the features of olive oil that are associated with tumour regression.
2. Materials and Methods
Chemicals and reagents - 7,12-Dimethylbenz[a]anthracene (DMBA), which was produced by Sigma-Aldrich in the United States of America and had the product number D3254-1G (CAS registration number: 57-97-6), lot number SLBX1136, and P code: 1002660800, was acquired from a scientific chemical supplier known as Patna, which is located in the state of Bihar in India. Each and every one of the remaining solvents and chemicals that were utilised were of an analytical grade, displaying a purity level of 99%.
The Production of Olive Oil: Commercial olive oil (Figaro) was procured from the market Batch number L4450U
Animals : There are twelve females in total. Rodents of the Charles Foster strain were obtained from the animal facility of the Mahavir Cancer Sansthan and Research Centre in Patna, India. In every instance of animal testing, the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), located in New Delhi, was responsible for establishing the guidelines and regulations that were followed. In accordance with the recommendations of the Institutional Animal Ethics Committee (IAEC), the experimental inquiry was given the go-ahead. The diets that were given to the rats consisted of drinking water and eating grain. During the seven days that before the beginning of the experimental procedures, rats were given the opportunity to become accustomed to the environment. Standard polypropylene enclosures were utilised to house the rats in the laboratory. Each enclosure included two animals, and the animals were randomly assigned to either the control group or the treatment group. The ambient temperature of the animal facility was kept at 22 ± 2 degrees Celsius for the rodents, and a light/dark cycle that lasted for approximately 12 hours was established.
Using Animals for Experimental Design: A total of twelve female Charles Foster strain rats, ranging in age from 55 to 60 days and weighing a total of 150 Η 10 g, were divided into three groups, with each group consisting of four animals. Group I was the control group, and Group II consisted of rats for whom DMBA was the only chemical administered. Olive oil was administered to the rats in Group III after they had been induced with DMBA. Administered olive oil 2000 milligrammes per kilogramme of body weight on a daily basis for a period of four weeks after the formation of the tumour (about half a centimetre). Rats were put under anaesthesia with diethyl ether at the completion of the dose regimen, and then they were put to sleep with diethyl ether before being put to sleep during the diestrous phase of the oestrous cycle. Blood samples from the hearts of the experimental rats were taken after they had been put to sleep through euthanasia. Following the isolation of serum, subsequent biochemical studies, evaluations of lipid peroxidation and tumour necrosis factor-alpha, as well as hormonal evaluations of oestrogen and progesterone, were carried out. Tissues from the breast were stored in formalin at a concentration of 10% for further histological examination.
Tumour Model : Inducing mammary gland tumours in female Charles Foster rats that were 55 days old and weighed between 150 and 10 grammes was the subject of this study. The mice were given a single, freshly manufactured dosage of DMBA (7,12-dimethylbenz(a)anthracene) that was diluted in maize oil and injected intragastrically using the gavage method. The dosage was 15 mg/mL. In the cohort, there were eight female rodents, all of them were 55 days old and weighed approximately 150 ± 10 grammes. Beginning in the fourth week after receiving DMBA treatment, rats were palpated on a weekly basis in order to track the growth of tumours over the course of the study. It was not until the sixteenth week after the injection of DMBA that the first signs of tumour formation were seen; by the eighteenth week, all eight animals had already displayed signs of tumour existence.
It was determined how much the mammary tumour was. On the basis of the size of the tumour, a Vernier calliper was utilised. Using the formula V(cm³) = (L × B²)/2, the volume of the tumour (V) was determined. In this formula, L represents the largest diameter, while B represents the smallest diameter. Both of these diameters were measured in centimetres (cm) and were orientated perpendicularly as the formula was used.
LPO stands for lipid peroxidation. The twofold heating method was utilised in order to ascertain whether or not thiobarbituric acid reactive chemicals (TBARS) were present. These substances are characteristics that are suggestive of lipid peroxidation (LPO). The spectrophotometric quantification of the colour that is produced as a result of the reaction between malondialdehyde (MDA) and thiobarbituric acid (TBA) is the foundation of this method. For the purpose of this experiment, 2.5 millilitres of a solution containing 10% trichloroacetic acid (TCA) was mixed with 0.5 millilitres of serum in a centrifuge tube. The mixture was then heated in a water bath at a temperature of 90 degrees Celsius for fifteen minutes. After bringing the mixture down to room temperature, it was centrifuged at a speed of three thousand revolutions per minute for ten minutes. Next, in a test tube, 2 millilitres of the supernatant were combined with 1 millilitre of a TBA solution that had a concentration of 0.675%. After that, the combination was heated for fifteen minutes in a water bath at a temperature of ninety degrees Celsius, and then it was cooled down to the temperature of the surrounding environment. At a wavelength of 532 nm, an absorbance measurement was performed using a UV-Vis spectrophotometer manufactured by Thermo Scientific in the United States of America (UV-10).
A hormonal analysis is performed. The ELISA technique was utilised in order to carry out the hormonal evaluation. Monobind Inc., which has its primary place of business located at 100 North Pointe Drive, Lake Forest, California 92630, United States of America, was the company that created the oestradiol and progesterone ELISA reagent. The concentrations of oestradiol (Lot No. EIA-49K2I8) and progesterone (Lot No. EIA-48K2E8) were determined in accordance with the instructions provided by the manufacturer and Saunders, 199439.
Following the calibration of the standard oestradiol range, aliquots of serum samples containing 25 μ5L were dispensed into the micro well plate. Oestradiol biotin reagent was put into each micro well in the form of an initial aliquot that was fifty millilitres in volume. After that, the microplate was gently stirred for twenty to thirty seconds, and then it was incubated at room temperature for approximately thirty minutes. In each well, fifty microlitres of oestradiol enzyme reagent was administered, and then the mixture was gently stirred for an additional twenty to thirty seconds. After that, the plate was covered and left to incubate at room temperature for a period of ninety straight minutes. After that, the contents of the micro well plate were removed by aspiration, followed by three washes with a total volume of fifty-five litres of wash buffer, and finally, blotting was performed. Following this, 100 u00b5L of substrate solution was added to each well, and the mixture was then incubated at room temperature for an additional twenty minutes. After a time of incubation, fifty millilitres of stop solution was later added to each well, and the mixture was carefully mixed. With the help of a Merck ELISA reader, absorbance was measured at 450 nm (with a reference wavelength of 620-630 nm), and the results are reported as pg/mL.
It is progesterone. At the same time as 25 μ5L aliquots of serum samples were distributed into the microwell plate, the reference range for progesterone that had been developed was utilised. A total volume of fifty millilitres of progesterone enzyme reagent was initially aliquoted and then subsequently distributed into each well of the microplate. Subsequent to the gentle agitation of the microwell plate for a duration of twenty to thirty seconds, fifty microlitres of progesterone biotin reagent was introduced into each individual well. Twenty to thirty seconds of slow agitation was performed on the microwell plate, after which it was covered and left to incubate at room temperature for sixty minutes. Aspiration was performed on the contents of the microwell plate, which was then followed by three treatments with 350 μL of wash buffer, and finally, blotting was performed. Following the dispensing of one hundred microlitres of substrate solution into each well, the mixture was allowed to incubate at room temperature for twenty minutes. After a time of incubation, fifty millilitres of stop solution was later added to each well, and the mixture was carefully mixed. Through the utilisation of a Merck ELISA reader, the absorbance was measured at 450 nm, with a reference wavelength ranging from 620 to 630 nm. The presentation of the results is in ng/mL.
The assay for tumour necrosis factor-alpha (TNF-α) detection. Utilising the ELISA technique, the levels of TNF-u03b1 in the serum were determined. Within the scope of this experiment, the rat TNF-u03b1 ELISA reagent, manufactured by Diaclone in France and bearing the catalogue number 872.010.001, was utilised. Evaluation of the serum levels of tumour necrosis factor-alpha was carried out in accordance with the instructions provided by the manufacturer. The capture antibody was initially pre-coated onto the microwell plate before the experiment began. One hundred millilitres of standard diluents and one hundred millilitres of serum were then put to the wells that had been designated to them. For the purpose of ensuring that each well received sufficient coverage, fifty microlitres of diluted detection antibody was applied to each well, and the wells were then incubated at room temperature for three hours. The material was discarded after it had been used, and then it was cleaned three times with a total volume of 300 ¼5L of laundry solution. After this, 100 u00b5L of streptavidin-HRP solution was dispensed into each well, and then the solution was incubated at room temperature for thirty minutes in order to ensure that all of the wells were completely covered. After that, the material was thrown away and washed three times with a total volume of 300 ¼5L of laundry solution. After this, 100 u00b5L of TMB substrate solution was added to each well, and the mixture was then incubated at room temperature for five to fifteen minutes in a covered environment that was shielded from light. At the conclusion of the incubation time, one hundred-five millilitres of stop reagent was applied to each well. An ELISA reader manufactured by Merck was used to measure absorbance at 450 nm (with a reference wavelength ranging from 620–630 nm), and the results are represented as pg/mL.
Analysis of statistical data - There is a presentation of the results in the form of the mean and the standard error of the mean (SEM). Through the utilisation of a two-way analysis of variance (ANOVA), the differences in tumour volume that were observed between the groups that were treated with DMBA and those that were treated with olive oil were evaluated. The independent variables that were included were time and treatment regimen. A one-way analysis of variance (ANOVA) was used to determine the statistical significance of differences between the control group and the treatment group for all of the other parameters. This was then followed by a Tukey post hoc test to determine whether or not there were multiple comparisons available. The finding was found to be statistically significant, according to the findings. Analyses were carried out with the assistance of the GraphPad Prism 5 software (GraphPad Software, Inc., San Diego, United States of America).
Examination of the histopathology results. Specimens of breast tissue were exposed to 10% formalin for a period of twenty-four hours. Immediately after the tissues were dehydrated with ethanol, they were then embedded in paraffin. In order to conduct a histological examination using light microscopy, slices of five micrometres in thickness were prepared and stained with haematoxylin and eosin.
The clearance of ethics. All international, national, and institutional regulations that were applicable for the treatment and utilisation of animals were adhered to. At the Mahavir Cancer Sansthan and Research Centre in Patna, India (CPCSEA Registration No. u2026u2026), the experimental inquiry was given the go-ahead by the Institutional Animal Ethics Committee (IAEC), which assigned the protocol number 2021/1F-06/10/21. In every instance of animal testing, the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), located in New Delhi, was responsible for establishing the guidelines and regulations that were followed. According to the writers, this article does not contain any investigations that require the participation of human beings.
3. Results
In the present investigation, the anticancer property of olive oil was evaluated on a DMBA-induced breast tumor model on Charles Foster rats. A 200 mg/kg dose of olive oil was administered to a DMBA-induced breast tumor model on rats, and the efficacy of olive oil was observed through tumor volume, lipid peroxidation (LPO) test, estradiol and progesterone levels, serum TNF-α level, and histopathological examination of mammary tissue. The following findings were observed (Figure 1-8).
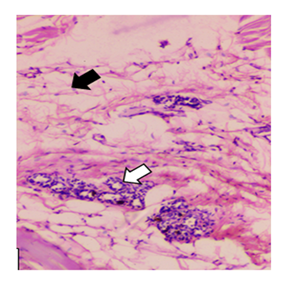
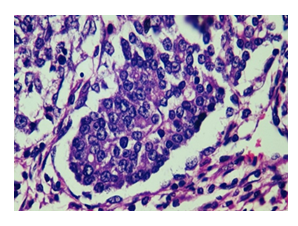
Figure 7: The image (H&E, 40x) shows high-grade mammary adenocarcinoma induced by DMBA. The presence of pleomorphism, high mitotic activity, necrosis and stromal invasion confirm the malignant nature of the lesion. Focal coagulative necrosis is present, particularly in central tumour areas, likely due to rapid proliferation outpacing blood supply. Apoptotic bodies are seen at the periphery of tumour lobules.
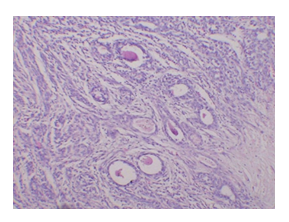
Figure 8: This image (H&E, 10x) shows a moderately differentiated adenocarcinoma of the mammary gland with partial evidence of therapeutic response. Apoptosis, reduced mitoses and inflammatory infiltration may reflect the anticancer potential of olive oil. The tumour architecture is still preserved but there is visible tumour regression in some foci a promising sign of therapeutic effect.
4. Discussion
In the present investigation after administration of olive oil for 4 weeks reduction in tumor size was observed from 1.2 cm to 0.6 cm which is a significant reduction. Reduction in tuomor size was associated with a significant decrease in LPO level which is marked by MDA levels. The decrease in MDA levels exhibit the inhibitor activity of olive oil in production of LPO levels. It means olive oil helps to reduce oxidative stress in the rats. The decreased serum tnf alpha level was also observed in my study which shows anti-inflammatory and anti-proliferative property of olive oil. Phytochemicals, primarily phenolic compounds (oleuropein, hydroxytyrosol, tyrosol) and triterpenic acids (oleanolic, maslinic acid) present in olive could be reason to observe reduction in tumor volume, LPO levels and tnf alpha levels were observed as compared to DMBA treated in the present study. On the other hand, therapeutic response of olive was also observed in our present histopathological study. Apoptosis, reduced mitoses and inflammatory infiltration may reflect the anticancer potential of olive oil. The tumour architecture is still preserved but there is visible tumour regression in some foci a promising sign of therapeutic effect. Olive oil contains phenolic compounds like oleuropein that can act as weak phytoestrogens, potentially mediating estrogen receptor activity and helps to maintain hormone level. It has been shown to offer protective, estrogen-like effects on bone health in low-estrogen states (e.g., menopause) and may help reduce breast cancer risk by modulating estrogen pathways. Hence, in our present study a decrease in level of estradiol was observed as compared to DMBA treated, however no changes in serum progesterone level was observed.
The contents of olive oil have been the subject of an increasing amount of study, which suggests that they may offer protection against the development of many types of cancer [9,10]. These findings are in line with the findings of other research that is relevant to the topic, and they are compatible with the conclusions of the present work.
When viewed from a mechanistic perspective, our findings are in agreement with a number of experimental studies that were carried out in vivo. In large part, the positive effects of olive oil can be related to its unusual composition, which is rich in monounsaturated fatty acids (mainly oleic acid) [11], squalene, and phenolic compounds (simple phenols, secoiridoids, and lignans) [8,12]. Olive oil is a rich source of these components. They have potent anti-oxidant characteristics that reduce the amount of cellular oxidative stress and DNA damage through a process known as scavenging. In addition, they have an effect on critical signalling pathways that are involved with the development of cancer [13]. The ability of oleic acid to block the overexpression of Her-2/neu, which is associated with breast cancer, as well as to upregulate PEA3, which is a transcriptional regulator of the HER2 gene, has been demonstrated by studies conducted in vitro [14]. Additionally, it has been found to suppress the production of the fatty acid synthase gene, which is a gene whose levels are frequently raised in breast cancers [15]. This is a significant discovery. It was revealed that hydroxytyrosol [16] and squalene [17] can reduce the amount of reactive oxygen species in human mammary epithelial cells (MCF10A) and protect against oxidative damage. This was found out by the utilisation of these two different chemicals. A higher positive effect was reported in an experimental model of breast cancer for mice who were fed a diet that was rich in olive oil as contrasted to a diet that was high in maize. This was the case when an experimental model of breast cancer was used. Furthermore, the tumours behaved in a manner that was less aggressive in these animals. According to [18], the underlying mechanisms involved the alteration of cellular membranes, signalling pathways, and gene expression, which ultimately led to a reduction in the amount of DNA damage, an increase in the amount of apoptosis, and a decrease in the amount of proliferation which is a fair level of agreement our present study.
5. Conclusion
The current study concludes that the use of olive oil, particularly extra virgin olive oil with its high content of olive polyphenols and lignans, is more effective than other oils that might be substituted when it comes to the health benefits of the Mediterranean diet, especially with regard to breast cancer protection. To keep all the benefits, it's also a good idea to try eating some of the extra virgin olive oil raw in addition to cooking. This is made simple by the abundance of Mediterranean dips and salad dressings made with olive oil, as well as the fact that high-quality extra virgin olive oil is a great sauce and condiment on its own.
Acknowledgements:
The authors are thankful to Cytopathologists, Mahavir Cancer Sansthan & Research Centre, Patna for this valuable inputs and support.
Funding :
The study was supported by Mahavir Cancer Institute itself.
Disclosure :
The authors declare that they have no conflicts of interest in regard to this study.
References
- Albuquerque RCR, Baltar VT, Marchioni DML. Breast cancer and dietary patterns: A systematic review. Nutr Rev 72 (2014): 1-17.
- Escrich E, Solanas M, Moral R, et al. Modulatory effects and molecular mechanisms of olive oil and other dietary lipids in breast cancer. Curr Pharm Des 17 (2011): 813-830.
- Escrich E, Solanas M, Moral R. Olive oil and other dietary lipids in cancer: Experimental approaches. Olive Oil Health (2006): 317-374.
- Lai H, Singh NP. Oral artemisinin prevents and delays the development of 7,12-dimethylbenz[a]anthracene (DMBA)-induced breast cancer in the rat. Cancer Lett 231 (2006): 43-48.
- Krishnamoorthy D, Sankaran M. Modulatory effect of Pleurotus ostreatus on oxidant/antioxidant status in 7,12-dimethylbenz(a)anthracene induced mammary carcinoma in experimental rats - A dose-response study. J Cancer Res Ther 12 (2016): 386-394.
- Barros ACSD, Muranaka ENK, Mori LJ, et al. Induction of experimental mammary carcinogenesis in rats with 7,12-dimethylbenz(a)anthracene. Rev Hosp Clin Fac Med Sao Paulo 59 (2004): 257-261.
- Hassan BAR. Medicinal plants: Use in prevention and treatment of diseases (2020).
- Owen RW, Giacosa A, Hull WE, et al. Olive-oil consumption and health: The possible role of antioxidants. Lancet Oncol 1 (2000): 107-112.
- Battino M, Forbes-Hernández TY, Gasparrini M, et al. Relevance of functional foods in the Mediterranean diet: The role of olive oil, berries and honey in the prevention of cancer and cardiovascular diseases. Crit Rev Food Sci Nutr 59 (2019): 893-920.
- Colomer R, Menéndez JA. Mediterranean diet, olive oil and cancer. Clin Transl Oncol 8 (2006): 15-21.
- Alarcón de la Lastra C, Barranco MD, Motilva V, et al. Mediterranean diet and health: Biological importance of olive oil. Curr Pharm Des 7 (2001): 933-950.
- Owen RW, Haubner R, Würtele G, et al. Olives and olive oil in cancer prevention. Eur J Cancer Prev 13 (2004): 319-326.
- Cicerale S, Conlan XA, Sinclair AJ, et al. Chemistry and health of olive oil phenolics. Crit Rev Food Sci Nutr 49 (2009): 218-236.
- Menendez JA, Papadimitropoulou A, Vellon L, et al. A genomic explanation connecting Mediterranean diet, olive oil and cancer: Oleic acid induces formation of inhibitory PEA3 transcription factor-PEA3 DNA binding site complexes at the Her-2/neu (erbB-2) oncogene promoter in breast, ovarian and stomach cancer cells. Eur J Cancer 42 (2006): 2425-2432.
- Menendez J, Lupu R. Mediterranean dietary traditions for the molecular treatment of human cancer: Anti-oncogenic actions of the main olive oil monounsaturated fatty acid oleic acid (18:1n-9). Curr Pharm Biotechnol 7 (2006): 495-502.
- Warleta F, Quesada CS, Campos M, et al. Hydroxytyrosol protects against oxidative DNA damage in human breast cells. Nutrients 3 (2011): 839-857.
- Warleta F, Campos M, Allouche Y, et al. Squalene protects against oxidative DNA damage in MCF10A human mammary epithelial cells but not in MCF7 and MDA-MB-231 human breast cancer cells. Food Chem Toxicol 48 (2010): 1092-1100.
- Escrich E, Moral R, Solanas M. Olive oil, an essential component of the Mediterranean diet, and breast cancer. Public Health Nutr 14 (2011): 2323-2332.


 Impact Factor:
* 5.8
Impact Factor:
* 5.8
 Acceptance Rate:
71.20%
Acceptance Rate:
71.20%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
