Quantitative Analysis of Foveal Avascular Zone (FAZ) Remodeling in Early Diabetic Retinopathy using AI-Powered OCT-Angiography Metrics
Sabah Lafta Ashash1*, Mohammed Kareem Mahmood Algburi2, Ali Adel Naseef Gassem3, Amar Adel Naseef Gassem3, Hassan alhajjaj4
1Introduktionslæge, Øjenafdelingen, Aalborg Universitets Hospital, Aalborg, Denmark
2Medical Department, Holbaek Hospital, Holbaek, Denmark
3Department of Medicine, Sjælland Universitets Hospital, Køge, , Denmark
4Bagdad Teaching Hospital, University of Baghdad, Baghdad, Iraq
*Corresponding Author: Sabah Lafta Ashash, Introduktionslæge, Øjenafdelingen, Aalborg Universitets Hospital, Aalborg, Denmark.
Received: 13 March 2026; Accepted: 20 March 2026; Published: 02 April 2026
Article Information
Citation: Sabah Lafta Ashash, Mohammed Kareem Mahmood Algburi, Ali Adel Naseef Gassem, Amar Adel Naseef Gassem, Hassan Alhajjaj. Quantitative Analysis of Foveal Avascular Zone (FAZ) Remodeling in Early Diabetic Retinopathy using AI-Powered OCTAngiography Metrics. Archives of Clinical and Biomedical Research. 10 (2026): 138-146.
View / Download Pdf Share at FacebookAbstract
Background: Diabetic Retinopathy (DR) is a major cause of vision loss. artificial intelligence powered optical coherence tomography angiography (OCTA) provides a non-invasive, quantitative method to detect early retinal ischemic changes by analyzing foveal avascular zone (FAZ) remodeling.
Objective: This study utilizes AI-powered OCTA metrics to assess FAZ remodeling and macular vessel density, evaluating their predictive value for early diabetic retinopathy and their association with visual function.
Methods: This prospective cross-sectional study evaluated 100 participants (50 with early diabetic retinopathy, 50 healthy controls). Using AI-based OCTA, we quantified FAZ morphology and macular vessel density. Statistical analysis, including multivariate logistic regression, assessed microvascular remodeling and identified independent predictive biomarkers for early disease detection.
Results: Early DR cases exhibited significantly larger FAZ area (0.41 ± 0.12 vs 0.27 ± 0.07 mm², P-value below 0.001), increased perimeter (2.68 ± 0.34 vs 2.05 ± 0.21 mm, P-value below 0.001), and higher acircularity index (1.29 ± 0.11 vs 1.08 ± 0.06, P P-value below 0.001). Macular vessel density in both superficial and deep capillary plexuses was significantly reduced in the DR group. FAZ enlargement correlated negatively with vessel density (r = −0.58 and −0.61, respectively; P-value below 0.001), and morphological distortion (acircularity/perimeter) correlated negatively with best-corrected visual acuity. Logistic regression confirmed FAZ metrics and reduced vessel density as independent predictors of early DR.
Conclusion: AI-based OCTA identifies significant FAZ enlargement and vessel density reduction in early DR. These metrics, particularly the acircularity index, are vital biomarkers for early detection and risk stratification.
Keywords
Early diabetic retinopathy; FAZ; OCTA; DR; AI-based OCTA
Article Details
1. Introduction
The prevalence of diabetic retinopathy (DR), an increasing retinal disease that is an important factor of vision impairment in people of working age, is predicted to rise over the coming years, reaching an estimated 160 million people by 2045 [1]. The main causes of visual loss throughout the course of DR are retinal neovascularization, diabetic macular ischemia (DMI) and diabetic macular edema (DME) [2]. Two primary classifications that is frequently employed for DR staging are Proliferative Diabetic Retinopathy (PDR) and Non-Proliferative Diabetic Retinopathy (NPDR) [3]. Making the distinction between PDR and NPDR can provide supplementary data for treatment guidance and disease progression prediction. Given the potential for serious side effects such vitreous hemorrhage and retinal detachment, PDR necessitates prompt intervention with anti-VEGF medication or laser therapy [4]. On the other hand, NPDR is treated with systemic management and routine monitoring, and it often has a lesser risk of vision loss [5]. Appropriate management to maintain eyesight is ensured by early differentiation [3].
An efficient retinal imaging technique, OCTA allows for non-invasive observation of the retinal microvasculature at different depths, which fluorescein angiography (FA) does not offer [6]. Given the significance of the macular region for vision, a number of imaging indicators have been described for the purpose of detecting macular changes in diabetic retinopathy and their potential diagnostic and prognostic applications [7-10].
High-acuity vision depends on FAZ, a small, capillary-free, elliptical or circular region in the center of the retina with distinct borders. It has been shown to be an important biomarker for evaluating retinal ischemia in a number of conditions, including DR [11]. A number of OCTA-derived metrics, including as FAZ area, perimeter, axis ratio, acircularity index, and roundness, have been developed to assess the foveal avascular zone morphology features in light of the enlargement and irregularity development in its boundaries prior to and through diabetic retinopathy advancement [12,13]. Numerous studies have shown that the foveal avascular zone changes improve the prediction accuracy of models for predicting progression in longitudinal studies [14,15], and are also associated with diabetic retinopathy severity and visual function [16,17]. The initial stage of the foveal avascular zone analysis, manual the foveal avascular zone segmentation, is labor-intensive, resource-intensive, subjective, and prone to human mistake. This is especially true when defining the foveal avascular zone with boundaries that are erratic, fading, or "moth-eaten," which are typical in diabetic retinopathy [9]. High inter-observer variability has resulted from this, especially in the deep capillary plexus [18-20]. Numerous studies have proposed automated methods for accurate FAZ in order to address these issues segmentation [21-25].
The combination of artificial intelligence and deep learning depend on segmentation algorithms into OCT-angiography platforms has significantly enhanced the precision, objectivity, and reproducibility of FAZ measurements. AI-powered quantitative metrics allow not only accurate calculation of FAZ area but also detailed morphological characterization through parameters such as acircularity and circularity indices, which may provide additional insight into microvascular remodeling beyond simple size measurements.
Understanding early FAZ remodeling and its relationship with vessel density and visual function may improve risk stratification and facilitate timely intervention in diabetic retinopathy. Therefore, the present study was designed to quantitatively analyze FAZ morphological alterations and macular vessel density variation in cases with early DR utilizing AI-powered OCT-angiography metrics, and to evaluate their associations with visual acuity and their potential role as independent predictors of early disease.
The objective of this study is to quantitatively assess foveal avascular zone (FAZ) remodeling in patients with early diabetic retinopathy (DR) using artificial intelligence-powered OCTA metrics. This achieved by comparing FAZ morphological parameters—including area, perimeter, acircularity index, and circularity index—as well as superficial and deep capillary plexus vessel density between early DR cases and healthy controls. Additionally, the study aims to evaluate the associations between these structural microvascular changes and their correlation with best-corrected visual acuity to measure functional impairment. Ultimately, the research sought to identify independent structural and microvascular predictors of early diabetic retinopathy through multivariate logistic regression analysis to enhance early disease detection.
2. Materials and Methods
This prospective cross-sectional comparative research was conducted from Mars 2025 to December 2025 and has been done to quantitatively evaluate FAZ remodeling and macular microvascular variations in cases with early DR utilizing artificial intelligence powered OCTA metrics. The research agreed with the principles of the Declaration of Helsinki [26] and received approval from the institutional ethics committee. Informed consent in writing was acquired from all subjects prior to enrollment.
A total of 100 eyes from 100 cases have been involved and divided into two groups: 50 eyes from cases diagnosed with early DR and fifty eyes from age- and sex-matched healthy controls. Early diabetic retinopathy was defined regarding the classification criteria established by the Early Treatment Diabetic Retinopathy Study, and only patients with mild non-proliferative DRwithout clinical data of diabetic macular edema were included [27].
2.1 Inclusion Criteria
Study Population and Eligibility
Participants were recruited based on specific inclusion and exclusion criteria. The Early Diabetic Retinopathy (DR) group included adults (≥ 18 years) with a confirmed diagnosis of Type 2 Diabetes Mellitus and mild non-proliferative DR, classified according to the Early Treatment Diabetic Retinopathy Study (ETDRS) scales. These patients were required to have an absence of clinically significant macular edema and clear ocular media allowing for high-quality OCTA imaging with a signal strength index meeting the manufacturer’s threshold.
The control group consisted of age- and sex-matched healthy individuals with no history of DM or evidence of retinal/optic nerve pathology.
2.2 Exclusion Criteria
Common exclusion criteria for both groups included moderate-to-severe NPDR, proliferative DR, or the presence of diabetic macular edema. Participants were also excluded if they had a history of retinal laser photocoagulation, vitreoretinal surgery, or any intraocular operation (excluding uncomplicated cataract extraction). Furthermore, eyes with coexisting retinal disorders (e.g., retinal vein occlusion, age-related macular degeneration, or glaucoma), high myopia (axial length > 26 mm or refractive error > −6.00 D), or media opacities were excluded. Finally, any OCTA images with significant motion artifacts, segmentation errors, or low signal strength were removed from the analysis.
2.3 Methods
All participants underwent comprehensive ophthalmologic investigation, including measurement of best-corrected visual acuity utilizing a standardized decimal visual acuity chart, slit-lamp biomicroscopy, intraocular pressure assessment, and investigation of dilated fundus. Best-corrected visual acuity values were stated in decimal units for statistical analysis.
Macular microvascular imaging has been conducted utilizing spectral-domain optical coherence tomography angiography with a standardized 3 × 3 millimeters scan protocol centered on the fovea. OCTA imaging utilized the RTVue XR 100 Avanti device (Version 2017.1.0.151, Optovue, Inc., Fremont, CA, USA) with 3 × 3 millimeters scans centered on the macula, acquired among 8:00 a.m. and 12:00 a.m. Superficial capillary plexus (SCP; from internal limiting membrane [ILM] to 9 μm above the inner plexiform layer [IPL]) and deep capillary plexus (DCP; 9 μm above IPL to 9 μm below outer plexiform layer [OPL]) slabs have been generated.
2.4 Quantitative Assessment and AI-Based Analysis
Quantitative assessment of the FAZ and vessel density has been conducted utilizing an artificial intelligence based automated segmentation algorithm integrated within the optical coherence tomography angiography platform. The algorithm employed a deep learning based convolutional neural network architecture designed for semantic segmentation of retinal vascular images.
Prior to segmentation, images underwent preprocessing procedures including motion correction, projection artifact removal (particularly for deep plexus images), grayscale normalization, and contrast enhancement to optimize vascular signal detection. The segmentation model utilized an encoder decoder framework, in which convolutional layers extracted multi-scale spatial features from the en face angiograms. These features enabled discrimination between perfused vascular structures and non-perfused regions. Pixel-wise classification was subsequently performed to identify the avascular area within the central macula. FAZ was defined as the largest contiguous non-flow region within the central foveal area.
Following segmentation, automated post-processing steps including morphological filtering, contour smoothing, and boundary refinement were applied to eliminate noise artifacts and ensure anatomical plausibility of the FAZ contour. All segmentations were visually reviewed, and scans with inaccurate boundary detection were excluded from further analysis.
From the delineated FAZ boundary, multiple morphological parameters were automatically computed. FAZ area (mm²) was calculated by summing the enclosed pixels and converting to square millimeters according to the image scaling factor. FAZ perimeter (mm) was derived from the total contour length. The acircularity index was calculated as the ratio between the calculated foveal avascular zone perimeter and the perimeter of a perfect circle with the same area, with values greater than 1 indicating increasing irregularity. The circularity index has been measured utilizing the formula 4π × (Area / Perimeter²), providing a normalized measure of shape regularity, with values approaching 1 indicating a more circular configuration. Vessel density analysis was performed separately for the deep and superficial capillary plexuses. The vascular maps were binarized using adaptive thresholding techniques to distinguish flow-positive pixels from background tissue. Vessel density has been known as the percentage area employed by perfused pixels within the predefined parafoveal region of interest. The foveal avascular zone area has been excluded from vessel density measurement to avoid artificial underestimation.
2.5 Statistical Analysis
Data has been collected by utilizing the Statistical Package for Social Science SPSS (IBM Corporation, Armonk, USA) version 26.0. Continuous variables have been stated as mean ± standard deviation, and categorical variables have been presented as incidences and percentages. Among-group comparisons have been performed utilizing independent samples t-tests for continuous variables and chi-square tests for categorical variables. Pearson association coefficient has been utilized to evaluate associations among FAZ parameters, vessel density metrics, and best-corrected visual acuity within the early DR group. Multivariate logistic regression analysis has been conducted to identify independent indicators of early diabetic retinopathy, with variables demonstrating statistical significance in univariate analysis entered into the regression model. A p-value below 0.05 has been recorded statistically significant.
3. Results
Regarding demographic data, there were insignificant distinctions between Early DR cases and controls in term of age or sex (P > 0.05), indicating good matching between groups. However, BCVA was significantly fewer in the Early DR group than controls (P-value below 0.001), suggesting early visual impairment associated with diabetic retinal changes as shown in Table 1.
|
Variable |
Early DR (n=50) |
Controls (n=50) |
P value |
|
|
Age (years) Mean± SD |
52.6 ± 6.4 |
50.9 ± 5.8 |
0.18 |
|
|
Sex |
Male |
28 (56%) |
26 (52%) |
0.69 |
|
Female |
22 (44%) |
24 (48%) |
||
|
BCVA (decimal) Mean± SD |
0.62 ± 0.18 |
0.92 ± 0.10 |
<0.001* |
|
BCVA: Best Corrected Visual Acuity
Table 1: Demographic data in the examined groups.
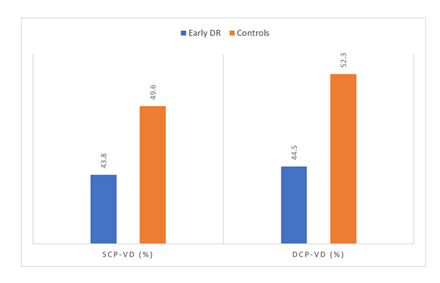
A significant reduction in macular vessel density in cases with early diabetic retinopathy than healthy controls. The mean superficial capillary plexus vessel density (SCP-VD) was significantly lower in the early diabetic retinopathy group (43.8 ± 3.9%) than in the control group (49.6 ± 3.1%) (P < 0.001), indicating early compromise of the superficial retinal microvasculature. Similarly, the deep capillary plexus vessel density (DCP-VD) was markedly reduced in patients with early diabetic retinopathy (44.5 ± 4.8%) compared to controls (52.3 ± 4.2%) (P-value equal to 0.001), as illustrated in Table 2 and Figure 1.
|
Parameter |
Early DR (n=50) |
Controls (n=50) |
P value |
|
SCP-VD (%) Mean± SD |
43.8 ± 3.9 |
49.6 ± 3.1 |
<0.001* |
|
DCP-VD (%) Mean± SD |
44.5 ± 4.8 |
52.3 ± 4.2 |
<0.001* |
Table 2: Vessel Density Analysis in the studied groups.
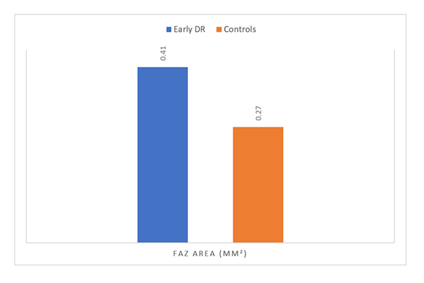
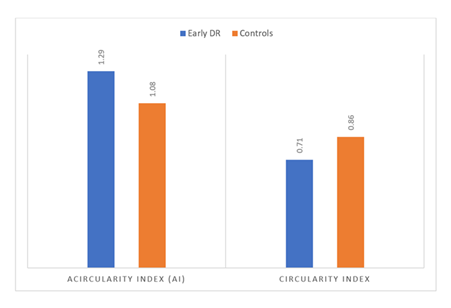
Our results demonstrated significant distinctions in FAZ morphological parameters among cases with early diabetic retinopathy and healthy controls as assessed by AI-based segmentation. The mean foveal avascular zone area was significantly higher in the early diabetic retinopathy group (0.41 ± 0.12 mm²) than controls (0.27 ± 0.07 mm²) (P-value equal to 0.001), indicating central macular capillary non-perfusion and early ischemic remodeling. Likewise, FAZ perimeter was significantly increased in the early diabetic retinopathy group (2.68 ± 0.34 mm) related to the control group (2.05 ± 0.21 mm) (P < 0.001), reflecting greater boundary irregularity and structural distortion of the perifoveal capillary arcade. The acircularity index was significantly higher in cases with early diabetic retinopathy (1.29 ± 0.11) than controls (1.08 ± 0.06) (P–value equal to 0.001), indicating increased deviation from a perfect circular configuration. Conversely, the circularity index was significantly fewer in the early diabetic retinopathy group (0.71 ± 0.08) than in controls (0.86 ± 0.05) (P < 0.001), confirming a loss of geometric regularity in the FAZ contour, as shown in Table 3, Figure 2 and Figure 3.
|
Parameter |
Early DR (n=50) |
Controls (n=50) |
P value |
|
FAZ Area (mm²) Mean± SD |
0.41 ± 0.12 |
0.27 ± 0.07 |
<0.001* |
|
FAZ Perimeter (mm) Mean± SD |
2.68 ± 0.34 |
2.05 ± 0.21 |
<0.001* |
|
Acircularity Index (AI) Mean± SD |
1.29 ± 0.11 |
1.08 ± 0.06 |
<0.001* |
|
Circularity Index Mean± SD |
0.71 ± 0.08 |
0.86 ± 0.05 |
<0.001* |
Table 3: FAZ Morphological Parameters (AI-based segmentation) in the Studied Groups.
There were significant associations among FAZ morphological parameters, vessel density metrics, and visual function in cases with early DR. A strong positive correlation has been observed among foveal avascular zone areas and FAZ perimeter (r = 0.81, P-value below 0.001), indicating that enlargement of the FAZ is accompanied by increased boundary length and structural expansion. Similarly, FAZ area showed a significant positive association with the acircularity index (r = 0.67, P < 0.001), suggesting that greater FAZ enlargement is associated with increased morphological irregularity. In addition, foveal avascular zone perimeter was strongly positively associated with the acircularity index (r = 0.74, P-value below 0.001), further confirming that shape distortion intensifies as the FAZ expands, as shown in Table 4.
|
Variable Pair |
r |
P value |
|
FAZ Area vs Perimeter |
0.81 |
<0.001** |
|
FAZ Area vs AI |
0.67 |
<0.001** |
|
FAZ Area vs SCP-VD |
-0.58 |
<0.001** |
|
FAZ Area vs DCP-VD |
-0.61 |
<0.001** |
|
Perimeter vs AI |
0.74 |
<0.001** |
|
AI vs BCVA |
-0.39 |
0.006* |
|
Perimeter vs BCVA |
-0.36 |
0.011* |
Table 4: Comparison among the FAZ (mm²), BCVA and other parameters among Early Diabetic Retinopathy.
Conversely, FAZ area demonstrated significant negative correlations with both superficial capillary plexus vessel density (r = −0.58, P-value below 0.001) and deep capillary plexus vessel density (r = −0.61, P-value below 0.001). These results demonstrate that increased central macular non-perfusion is closely associated with progressive microvascular rarefaction in both retinal plexuses. Regarding functional correlation, the acircularity index showed a moderate but significant negative association with best-corrected visual acuity (r = −0.39, P-value equal to 0.006), while FAZ perimeter also associated with negatively with visual acuity (r = −0.36, P-value equal to .011). These results suggest that greater FAZ distortion and irregularity are associated with early visual impairment (Table 4).
According to multivariate logistic regression analysis, FAZ morphological parameters demonstrated strong and statistically significant associations with early diabetic retinopathy. FAZ area demonstrated a significant positive relation (β = 1.84, P-value equal to 0.003), indicating that increasing FAZ enlargement significantly increases the likelihood of early disease. Similarly, FAZ perimeter was independently associated with early diabetic retinopathy (β = 2.21, P = 0.002), reflecting the contribution of boundary expansion and structural distortion to disease presence. Notably, the acircularity index emerged as the strongest morphological predictor (β = 3.46, P = 0.001), suggesting that FAZ shape irregularity may be a especially sensitive marker of early microvascular remodeling (Table 5).
In contrast, vessel density parameters showed significant negative associations. Superficial capillary plexus vessel density (β = −0.29, P-value equal to 0.004) and deep capillary plexus vessel density (β = −0.24, P-value equal to 0.006) were independently and inversely related to early diabetic retinopathy, indicating that reductions in macular perfusion significantly increase disease probability. These findings demonstrated that the regression model confirmed that both FAZ morphological distortion and reduced retinal vessel density serve as independent and complementary predictors of early diabetic retinopathy, underscoring their potential utilization as quantitative biomarkers for initial diagnosis and risk stratification as shown in Table 5.
|
Variable |
β |
P value |
|
FAZ Area |
1.84 |
0.003* |
|
FAZ Perimeter |
2.21 |
0.002* |
|
Acircularity Index |
3.46 |
0.001** |
|
SCP-VD |
-0.29 |
0.004* |
|
DCP-VD |
-0.24 |
0.006* |
Table 5: Multivariate Logistic Regression Examination Determining Autonomous indicators of Early Diabetic Retinopathy.
4. Discussion
The examination of the foveal avascular zone is essential for diagnosing and monitoring numerous ocular illnesses. Due to its central position in the fovea, a region essential to high-acuity vision, morphological variations in FAZ have been correlated with pathological disorders involving DR, macular edema, and age-related macular degeneration (AMD). The variations indicate essential pathogenic mechanisms, such as tissue hypoxia, inflammation, and vascular remodeling, thereby underscoring the significance of the foveal avascular zone as a clinical and prognostic indicator [28]. The quality of images in OCT-A tests is a crucial factor impacting segmentation accuracy, as artifacts, media opacities, and variations in acquisition can prevent the accurate diagnosis of vascular structures. The 2020 research has shown that ocular media opacities significantly influence vascular density (VD) results across several OCT-A devices, underscoring the differing sensitivities of segmentation algorithms to this interference. Manual foveal avascular zone segmentation is recognized for its exceptional precision and is commonly employed as a reference standard in ocular research. Nonetheless, its scalability is restricted due to the necessity for skilled professionals, the labor-intensive nature of the procedure, and the possibility of interobserver variability.
In the current study, AI-powered quantitative OCTA revealed significant remodeling of FAZ and macular microvascular alterations in early diabetic retinopathy than age-matched healthy controls. Early diabetic eyes exhibited larger FAZ area and perimeter, increased acircularity index, and decreased circularity index, indicative of non-perfused capillary loss and structural distortion of the central macula. These alterations reflect underlying ischemia and capillary dropout, consistent with the early microangiopathic changes reported in OCTA analyses of diabetic patients.
Similarly, our results agreed with the study done by Sijilmassi [29]which assessed The size and morphology of the foveal avascular zone in cases with type 2 DM compared to healthy participants. The findings indicated that the foveal avascular zone area was markedly greater in diabetic eyes (mean = 0.50 millimeters) than in control eyes (mean = 0.37 millimeters), with a p-value of less than 0.01. The maximum and minimum diameters of the foveal avascular zone were significantly higher in the diabetes groups than the control group. The parameters related to the morphology of the foveal avascular zone were markedly reduced in the diabetic groups than the control group, with the exception of the axial ratio. The key finding of their investigation was that diabetic eyes lacking clinically observable diabetic retinopathy display morphological alterations and abnormalities at the foveal avascular zone boundary. Kim MIG et al. [30] examined the foveal avascular zone area utilizing OCTA in diabetic eyes with and without diabetic retinopathy (DR), discovering that the FAZ area was larger in cases without and in diabetic cases exhibiting different levels of DR. The expansion of the FAZ in diabetic cases is explained by retinal capillary obstruction and the deterioration of vascular integrity, indicating that microvascular dysfunction is present prior to the clinical manifestation of diabetic retinopathy [28,31].
The observed FAZ enlargement and irregularity align with recent findings demonstrating that FAZ morphology correlates with visual impairment and disease severity. The study done by Duffy et al. [32] reported that FAZ enlargement correlates with declines in best-corrected and low-luminance visual acuity across a broad spectrum of diabetic retinopathy severity, supporting the clinical relevance of detailed FAZ assessment beyond simple area measurement. Their results demonstrated that FAZ enlargement metrics were significantly correlated with reduced function of vision, reinforcing the idea that FAZ remodeling reflects functional ischemia in diabetic eyes.
Our results showed that vessel density metrics in both the deep capillary plexus (DCP-VD) and superficial capillary plexus (SCP-VD) were significantly reduced in early diabetic retinopathy. These findings corroborate previous OCTA studies demonstrating microvascular rarefaction before the development of clinically advanced disease. Yao et al. [33]demonstrated that diabetic patients exhibited significant reductions in vessel density on OCTA, highlighting that both superficial and deep capillary networks undergo early perfusion loss. Additionally, Tang et al. [34]employed deep learning segmentation to analyze retinal vessel metrics in type 1 diabetes. Their findings revealed that reduced macular vessel density was observed even in early non-proliferative stages, further supporting the utility of quantitative OCTA metrics in early diabetic microvascular assessment.
The strong negative correlations observed between foveal avascular zone area and both SCP-VD and DCP-VD in our cohort indicate that FAZ remodeling is closely linked to macular capillary rarefaction. This relationship aligns with recent reports suggesting that statistical models incorporating FAZ enlargement alongside vessel density metrics improve detection and stratification of diabetic retinopathy severity. The study done by Busza et al. [35] showed that FAZ enlargement enhances statistical models for detecting referable diabetic retinopathy and proliferative disease when combined with OCTA parameters such as geometric perfusion deficit and vessel density, emphasizing the incremental value of detailed FAZ metrics.
The use of AI-based segmentation is increasingly recognized as a robust tool for objective and reproducible FAZ analysis. Semantic segmentation models have demonstrated high accuracy and strong agreement with manual annotations across diverse cohorts, enhancing the precision of quantitative OCTA measurements. A recent cross-sectional study by Carvalho et al. [36] aimed to evaluate the semantic segmentation algorithms for FAZ delineation. They reported high overlapping metrics and highlighted their potential to standardize FAZ analysis in clinical and research settings, reducing variability inherent to manual grading. In our cohort, increased FAZ distortion as reflected by acircularity and perimeter was negatively related with best-corrected visual acuity in early DR. This observation was parallel with previous study by Duffy et al. [32] indicating that structural ischemic changes in the macula precede and predict functional visual decline, underscoring the importance of detailed morphological characterization of the FAZ in addition to vessel density quantification.
In summary, these findings support the hypothesis that early diabetic retinopathy is characterized by both macular perfusion loss and significant FAZ remodeling, measurable using high-resolution, AI-enhanced OCTA analysis. The integration of detailed FAZ metrics and vessel density parameters may provide a more sensitive biomarker profile for early diabetic microvascular damage than either measure alone.
5. Conclusion
This study concluded that AI-powered quantitative optical coherence tomography angiography analysis demonstrated significant FAZ remodeling and reduced macular vessel density in early DR. Increase and morphological distortion of FAZ particularly increased acircularity are strongly associated with reduced vessel density and early visual impairment. The foveal avascular zone morphological parameters and vessel density metrics serve as independent predictors of early diabetic retinal microvascular damage. Artificial intelligence-based segmentation enhances precision and reproducibility, supporting its potential role as a reliable, non-invasive indicator for early diagnosis, disease monitoring and possibility stratification, Future longitudinal investigations are warranted to validate AI-derived FAZ metrics as predictive tools for disease progression and to integrate these quantitative biomarkers into standardized diabetic retinopathy screening protocols.
6. Author contributions
Conceptualization, Sabah Lafta Ashash, Mohammed Kareem Mahmood Algburi, Ali Adel Naseef Gassem, Amar Adel Naseef Gassem, Hassan alhajjaj; Formal analysis, Sabah Lafta Ashash, Amar Adel Naseef Gassem, Hassan alhajjaj; Investigation, Sabah Lafta Ashash, Hassan alhajjaj Hamied; Project administration, Sabah Lafta Ashash, Mohammed Kareem Mahmood Algburi, Ali Adel Naseef Gassem, Amar Adel Naseef Gassem, Hassan alhajjaj; Software, Sabah Lafta Ashash, Mohammed Kareem Mahmood Algburi, Ali Adel Naseef Gassem; Validation, Sabah Lafta Ashash, Mohammed Kareem Mahmood Algburi, Hassan alhajjaj Hamied; Formal analysis, Sabah Lafta Ashash, Mohammed Kareem Mahmood Algburi, Ali Adel Naseef Gassem; Visualization, Sabah Lafta Ashash, Hassan alhajjaj; Writing—original draft, Sabah Lafta Ashash; Writing—review, editing, and provided critical feedback to help shape the research, analysis, and manuscript, Sabah Lafta Ashash; All authors have read and agreed to the published version of the manuscript.
7. Declarations
Competing interests: The authors declare no competing interests.
Consent to participate: Not applicable.
Consent for Publication: Not applicable
Ethics Approval: The research agreed with the principles of the Declaration of Helsinki [26] and received approval from the institutional ethics committee. Informed consent in writing was acquired from all subjects prior to enrollment
Acknowledgments: NA
Funding: NA
Availability of data and materials: The data sets used in the current study are available from the corresponding author on reasonable request.
References
- Teo ZL, Tham Y-C, Yu M, et al. Global prevalence of diabetic retinopathy and projection of burden through 2045: systematic review and meta-analysis. Ophthalmology 128 (2021): 1580-1591.
- Cheung CMG, Pearce E, Fenner B, et al. Looking ahead: visual and anatomical endpoints in future trials of diabetic macular ischemia. Ophthalmologica 244 (2021): 451-464.
- Yang Z, Tan T-E, Shao Y, et al. Classification of diabetic retinopathy: Past, present and future. Frontiers in Endocrinology 13 (2022): 1079217.
- Gross JG, Glassman AR, Liu D, et al. Five-year outcomes of panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA Ophthalmology 136 (2018): 1138-1148.
- Chong DD, Das N, Singh RP. Diabetic retinopathy: Screening, prevention, and treatment. Cleveland Clinic Journal of Medicine 91 (2024): 503-510.
- Spaide RF, Fujimoto JG, Waheed NK, et al. Optical coherence tomography angiography. Progress in Retinal and Eye Research 64 (2018): 1-55.
- Riazi-Esfahani H, Jafari B, Azimi H, et al. Assessment of area and structural irregularity of retinal layers in diabetic retinopathy using machine learning and image processing techniques. Scientific Reports 14 (2024): 4013.
- Namvar E, Ahmadieh H, Maleki A, et al. Sensitivity and specificity of optical coherence tomography angiography for diagnosis and classification of diabetic retinopathy: a systematic review and meta-analysis. European Journal of Ophthalmology 33 (2023): 2068-2078.
- Waheed NK, Rosen RB, Jia Y, et al. Optical coherence tomography angiography in diabetic retinopathy. Progress in Retinal and Eye Research 97 (2023): 101206.
- Khalili Pour E, Rezaee K, Azimi H, et al. Automated machine learning-based classification of proliferative and non-proliferative diabetic retinopathy using optical coherence tomography angiography vascular density maps. Graefe's Archive for Clinical and Experimental Ophthalmology 261 (2023): 391-399.
- Conrath J, Giorgi R, Raccah D, et al. Foveal avascular zone in diabetic retinopathy: quantitative vs qualitative assessment. Eye 19 (2005): 322-326.
- Mirshahi R, Riazi-Esfahani H, Khalili Pour E, et al. Differentiating features of OCT angiography in diabetic macular edema. Scientific Reports 11 (2021): 23398.
- Kim K, Kim ES, Yu S-Y. Optical coherence tomography angiography analysis of foveal microvascular changes and inner retinal layer thinning in patients with diabetes. British Journal of Ophthalmology 102 (2018): 1226-1231.
- Yang D, Tang Z, Ran A, et al. Assessment of parafoveal diabetic macular ischemia on optical coherence tomography angiography images to predict diabetic retinal disease progression and visual acuity deterioration. JAMA Ophthalmology 141 (2023): 641-649.
- Sun Z, Tang F, Wong R, et al. OCT angiography metrics predict progression of diabetic retinopathy and development of diabetic macular edema: a prospective study. Ophthalmology 126 (2019): 1675-1684.
- Pan F, Banerjee S, Luk KS, et al. Optical defocus changes signaling of ganglion cells in the mouse retina. Investigative Ophthalmology & Visual Science 59 (2018): 706-706.
- Tsai AS, Jordan-Yu JM, Gan AT, et al. Diabetic macular ischemia: influence of optical coherence tomography angiography parameters on changes in functional outcomes over one year. Investigative Ophthalmology & Visual Science 62 (2021): 9-9.
- Coscas F, Sellam A, Glacet-Bernard A, et al. Normative data for vascular density in superficial and deep capillary plexuses of healthy adults assessed by optical coherence tomography angiography. Investigative Ophthalmology & Visual Science 57 (2016): OCT211-OCT223.
- Linderman RE, Muthiah MN, Omoba SB, et al. Variability of foveal avascular zone metrics derived from optical coherence tomography angiography images. Translational Vision Science & Technology 7 (2018): 20-20.
- Shahlaee A, Pefkianaki M, Hsu J, et al. Measurement of foveal avascular zone dimensions and its reliability in healthy eyes using optical coherence tomography angiography. American Journal of Ophthalmology 161 (2016): 50-55.
- Eladawi N, Elmogy M, Khalifa F, et al. Early diabetic retinopathy diagnosis based on local retinal blood vessel analysis in optical coherence tomography angiography (OCTA) images. Medical Physics 45 (2018): 4582-4599.
- Lu Y, Simonett JM, Wang J, et al. Evaluation of automatically quantified foveal avascular zone metrics for diagnosis of diabetic retinopathy using optical coherence tomography angiography. Investigative Ophthalmology & Visual Science 59 (2018): 2212-2221.
- Guo M, Zhao M, Cheong AM, et al. Automatic quantification of superficial foveal avascular zone in optical coherence tomography angiography implemented with deep learning. Visual Computing for Industry, Biomedicine, and Art 2 (2019): 21.
- Díaz M, Novo J, Cutrín P, et al. Automatic segmentation of the foveal avascular zone in ophthalmological OCT-A images. PLoS One 14 (2019): e0212364.
- Carmona EJ, Díaz M, Novo J, et al. Modeling, localization, and segmentation of the foveal avascular zone on retinal OCT-angiography images. IEEE Access 8 (2020): 152223-152238.
- Association WM. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310 (2013): 2191-2194.
- Wilkinson CP, Ferris III FL, Klein RE, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology 110 (2003): 1677-1682.
- Takase N, Nozaki M, Kato A, et al. Enlargement of foveal avascular zone in diabetic eyes evaluated by en face optical coherence tomography angiography. Retina 35 (2015): 2377-2383.
- Sijilmassi O. Quantitative analysis of different foveal avascular zone metrics in healthy and diabetic subjects. Diabetology 5 (2024): 246-254.
- Kim MIG, Lee MJE. Optical coherence tomography-angiography: comparison of the foveal avascular zone between diabetic retinopathy and normal subjects. Journal of the Korean Ophthalmological Society 58 (2017): 952-959.
- Chen Q, Ma Q, Wu C, et al. Macular vascular fractal dimension in the deep capillary layer as an early indicator of microvascular loss for retinopathy in type 2 diabetic patients. Investigative Ophthalmology & Visual Science 58 (2017): 3785-3794.
- Duffy BV, Castellanos-Canales D, Decker NL, et al. Foveal avascular zone enlargement correlates with visual acuity decline in patients with diabetic retinopathy. Ophthalmology Retina 9 (2025): 667-676.
- Yao Y, Wang Q, Yang J, et al. Associations of retinal microvascular alterations with diabetes mellitus: an OCTA-based cross-sectional study. BMC Ophthalmology 24 (2024): 245.
- Tang N, Su N, Zhang Z, et al. Retinal vessel metric analysis of type 1 diabetes mellitus in OCT angiography. Frontiers in Medicine 12 (2025): 1562809.
- Busza AM, Duffy BV, Kakihara S, et al. Foveal avascular zone enlargement significantly improves statistical models for detection of diabetic retinopathy severity. Ophthalmology Retina (2025).
- Carvalho BFdS, Rosa AAM, Scherer R, et al. Semantic segmentation of the avascular zone of the fovea in optical coherence tomography angiography: evaluation of techniques and applications in ocular diseases. International Journal of Retina and Vitreous 11 (2025): 109.


 Impact Factor:
* 5.8
Impact Factor:
* 5.8
 Acceptance Rate:
71.20%
Acceptance Rate:
71.20%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
